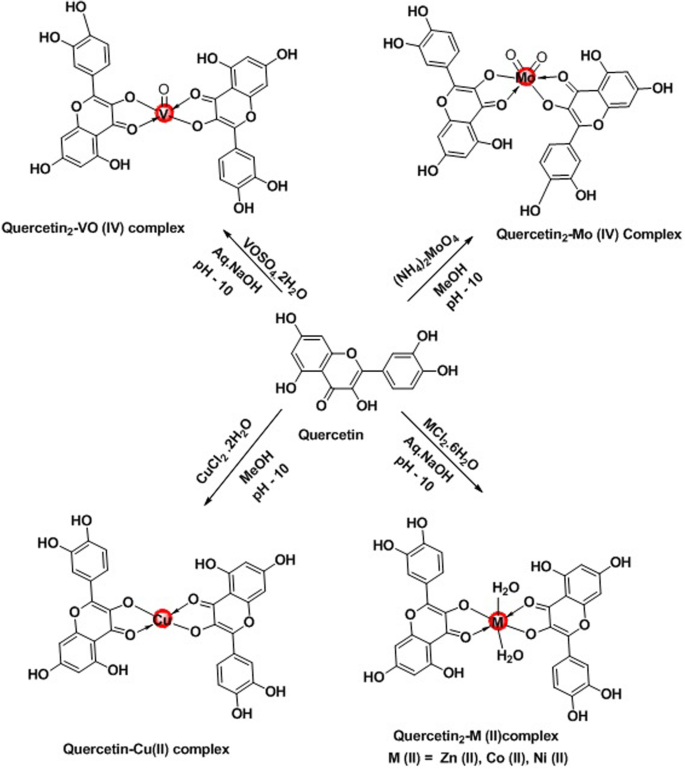
Synthesis, characterization, and pharmacological evaluation of some metal complexes of quercetin as P-gp inhibitors | Future Journal of Pharmaceutical Sciences | Full Text

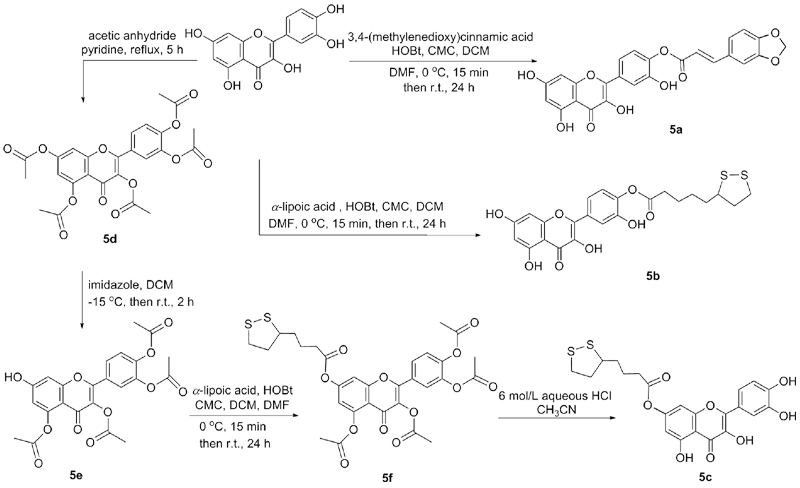
Synthesis and Testing of Novel Isomeric Mitochondriotropic Derivatives of Resveratrol and Quercetin | SpringerLink

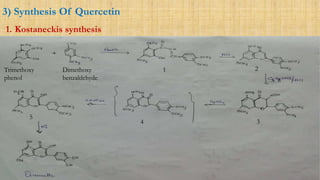
Scheme 11: Synthesis of fisetin (67) and quercetin (68). Reagents and... | Download Scientific Diagram

Biocatalytic Synthesis of Quercetin 3‐O‐Glucoside‐7‐O‐Rhamnoside by Metabolic Engineering of Escherichia coli - Roepke - 2013 - ChemBioChem - Wiley Online Library
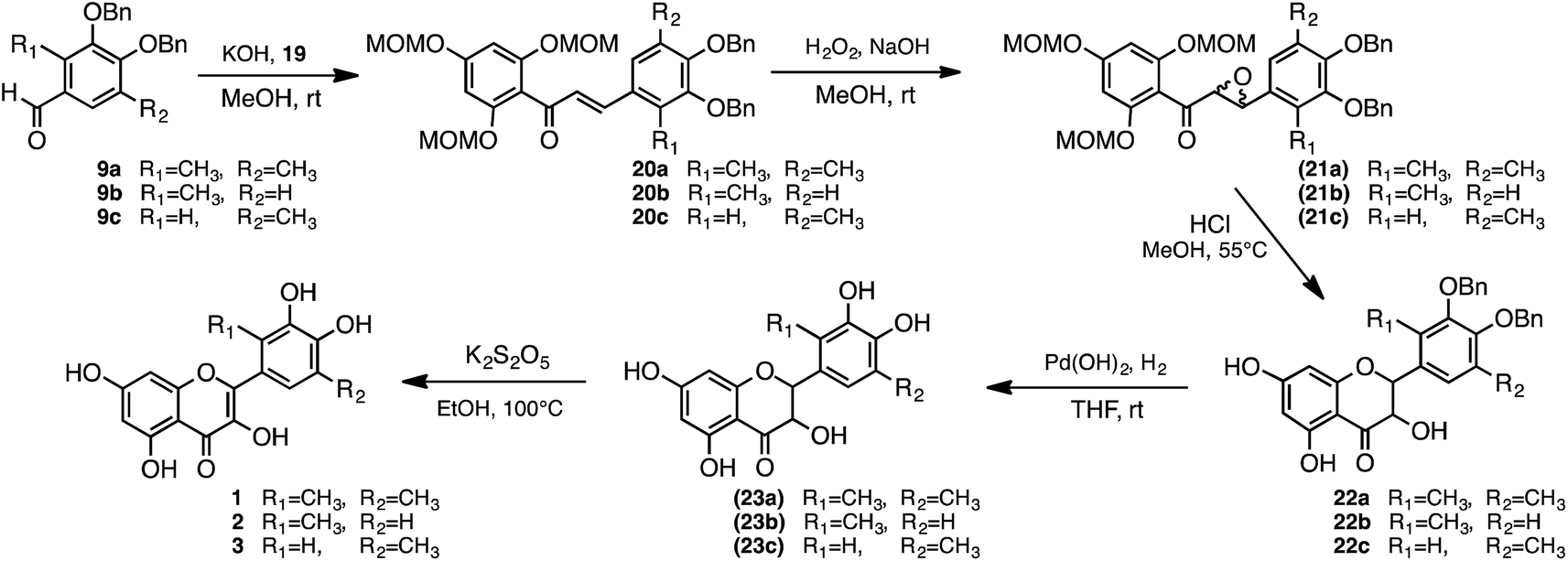
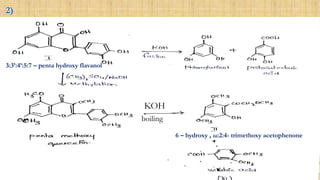
Synthesis of methylated quercetin analogues for enhancement of radical-scavenging activity - RSC Advances (RSC Publishing) DOI:10.1039/C7RA02329D

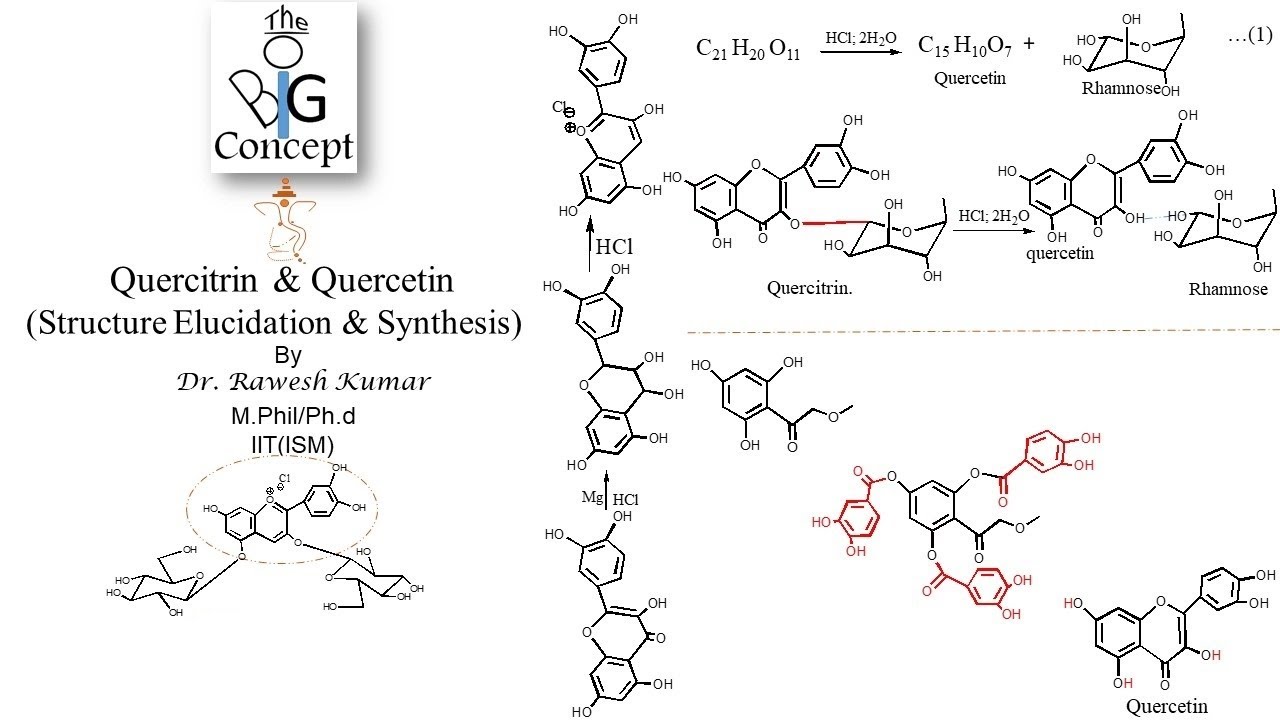
Figure 3 from Therapeutic charm of quercetin and its derivatives: a review of research and patents. | Semantic Scholar
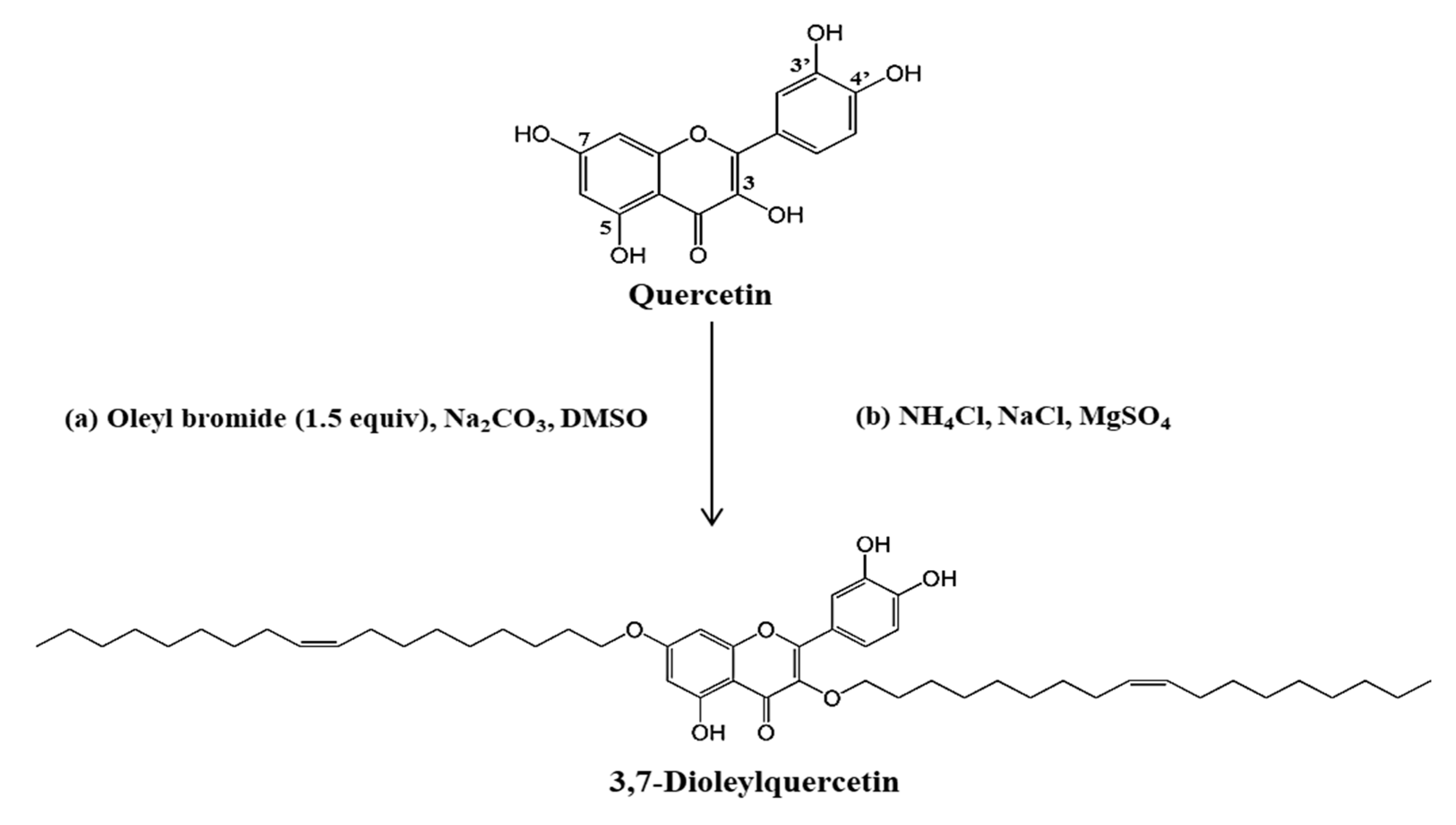
IJMS | Free Full-Text | Novel Quercetin Derivative of 3,7-Dioleylquercetin Shows Less Toxicity and Highly Potent Tyrosinase Inhibition Activity

Scheme 1 Synthesis of quercetin-alkylphospholipid conjugates I and II. | Download Scientific Diagram
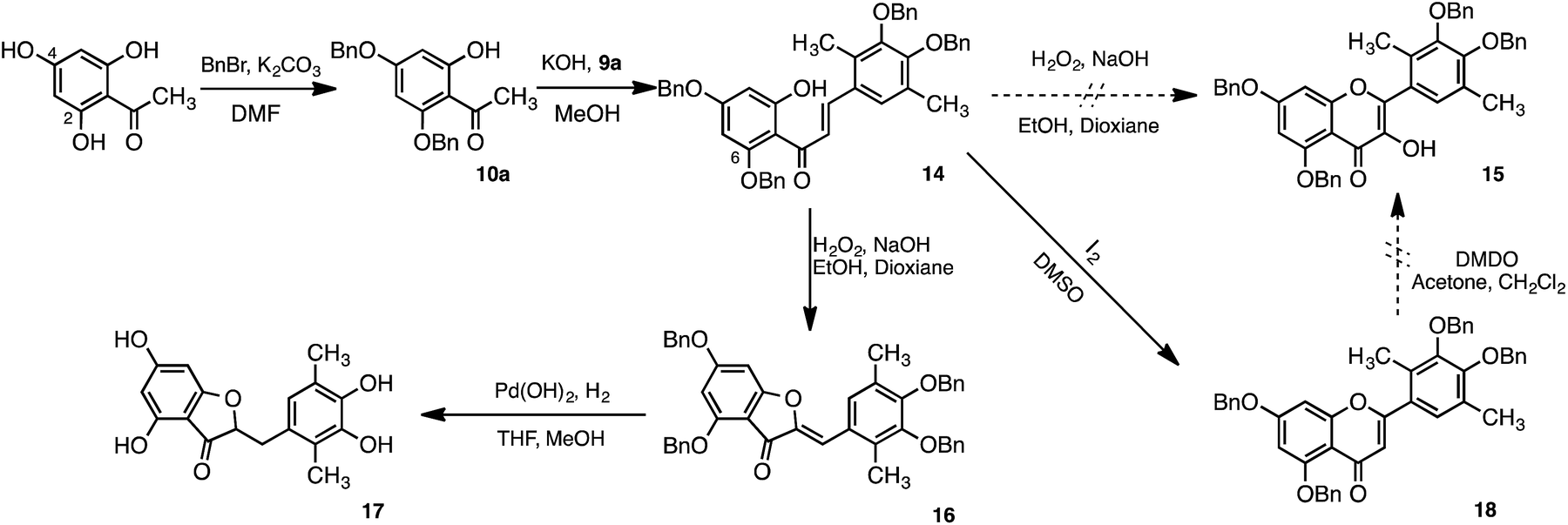
Synthesis of methylated quercetin analogues for enhancement of radical-scavenging activity - RSC Advances (RSC Publishing) DOI:10.1039/C7RA02329D

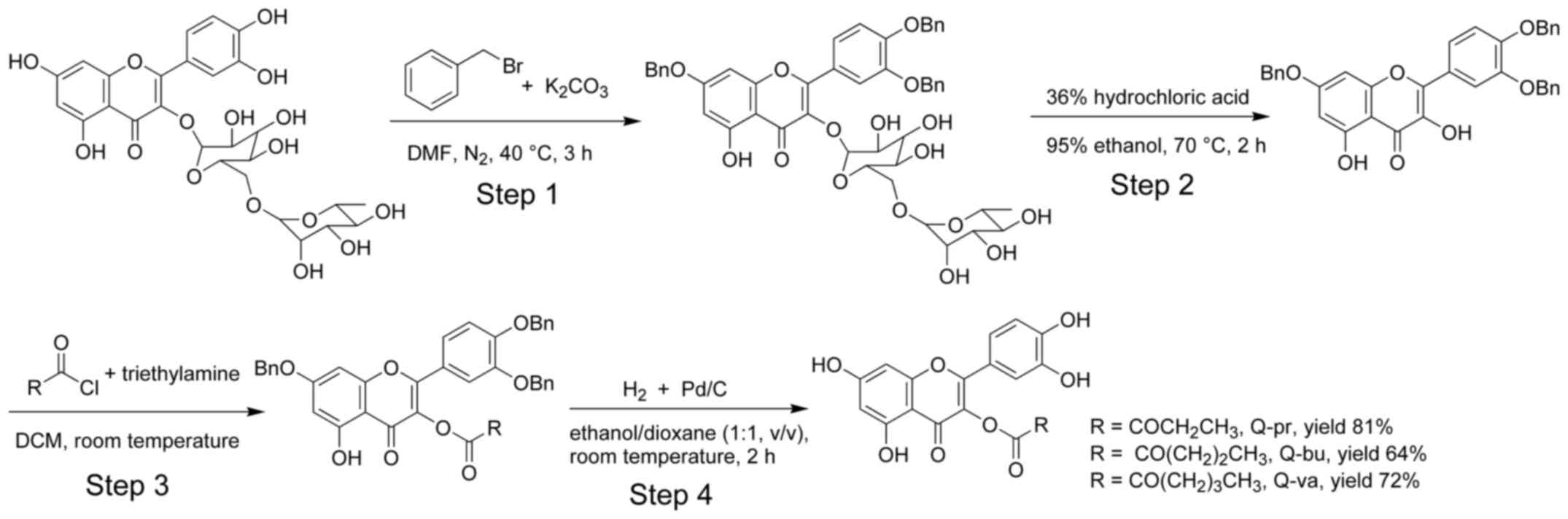
Molecules | Free Full-Text | Regioselective O-Derivatization of Quercetin via Ester Intermediates. An Improved Synthesis of Rhamnetin and Development of a New Mitochondriotropic Derivative

Synthesis of quercetin glycosides and their melanogenesis stimulatory activity in B16 melanoma cells - ScienceDirect

Effects of Functional Groups and Sugar Composition of Quercetin Derivatives on Their Radical Scavenging Properties | Journal of Natural Products
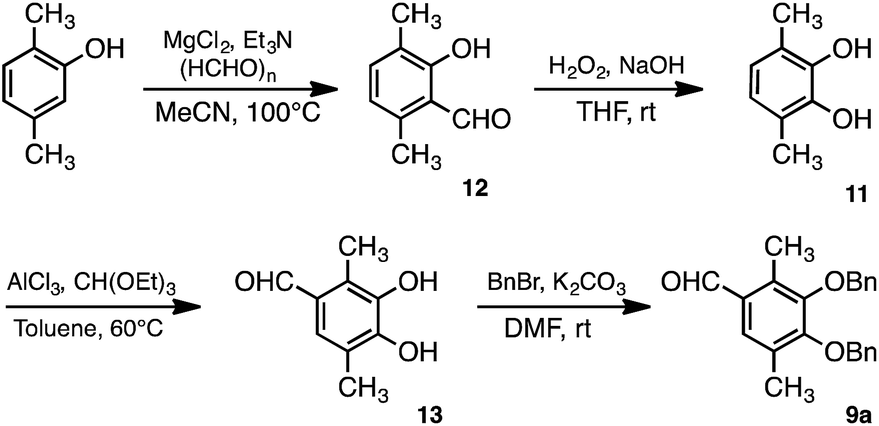
Synthesis of methylated quercetin analogues for enhancement of radical-scavenging activity - RSC Advances (RSC Publishing) DOI:10.1039/C7RA02329D