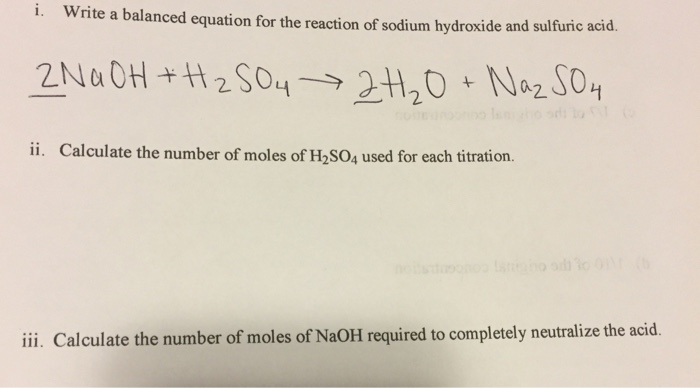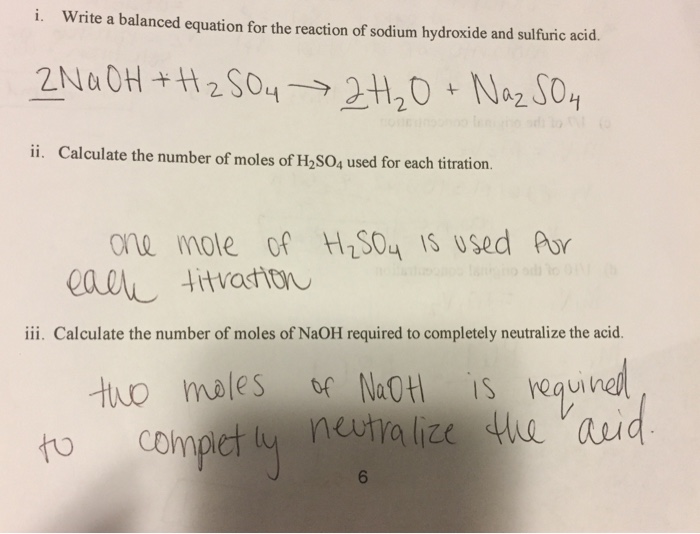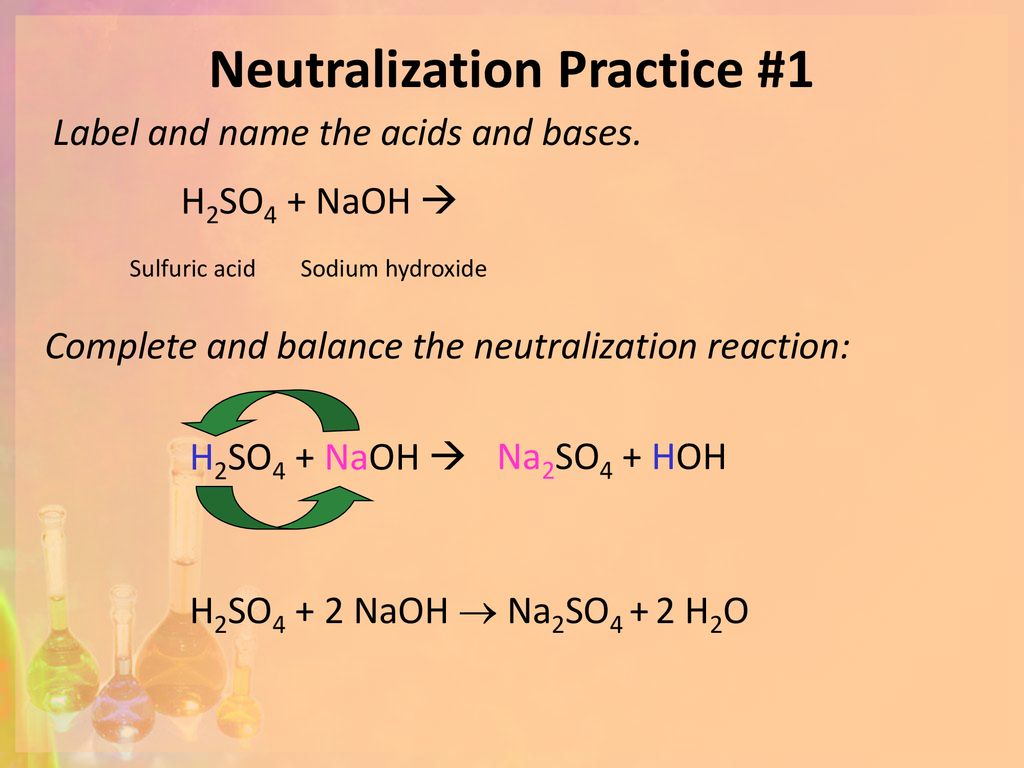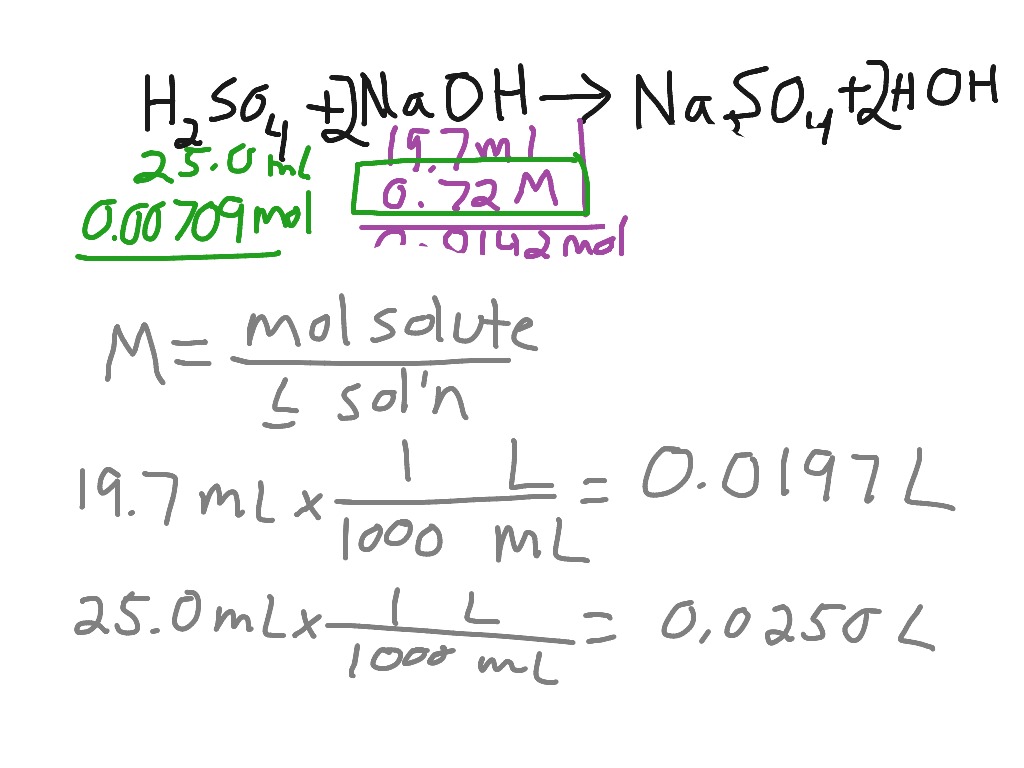
Titration of sulfuric acid with sodium hydroxide | Chemistry, Acids and Bases, Stoichiometry | ShowMe
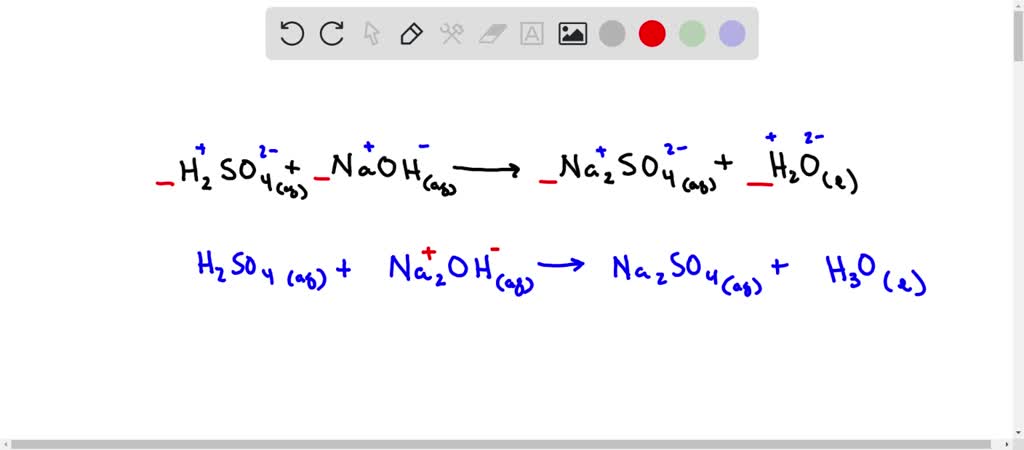
SOLVED: Sulfuric acid, H2SO4, can be neutralized by sodium hydroxide, NaOH. The unbalanced equation is: H2SO4(aq) + NaOH(aq) â†' Na2SO4(aq) + H2O(l). A student who was asked to balance the reaction wrote

Q How balance this equation H2SO4+NaOH→Na2SO4+H2O - Science - Chemical Reactions and Equations - 12449733 | Meritnation.com
![SOLVED: 1] Complete and balance the following neutralization equation. H2SO4 + NaOH ⟶⟶ + + ⟶⟶ BaCl2 + H2O + 3KOH ⟶⟶ K3PO4 + 2] How is a neutralization reaction related to a saponification reaction? SOLVED: 1] Complete and balance the following neutralization equation. H2SO4 + NaOH ⟶⟶ + + ⟶⟶ BaCl2 + H2O + 3KOH ⟶⟶ K3PO4 + 2] How is a neutralization reaction related to a saponification reaction?](https://cdn.numerade.com/ask_previews/8ccf65fd-0ce0-4618-a761-c39a7457fe1f_large.jpg)
SOLVED: 1] Complete and balance the following neutralization equation. H2SO4 + NaOH ⟶⟶ + + ⟶⟶ BaCl2 + H2O + 3KOH ⟶⟶ K3PO4 + 2] How is a neutralization reaction related to a saponification reaction?

Consider this skeleton equation: NaOH + H2SO4 → Na2SO4 + H2O Which of the following is the correct, - brainly.com